What is the process validation?
Process validation is the collection of data and documents that prove critical steps in the manufacturing process are stable and repeatable to ensure that the process, products, and devices are of consistent quality.
Forms of the process validation
- Prospective Validation: establishing documented evidence showing that the process, systems, devices, or process implementation meet the standards approved in the validation protocol.
- Retrospective Validation: is the process of validating marketed products based upon accumulated production, testing, and control data.
- Concurrent Validation: The appraisal is carried out concurrently with the production process of products to be circulated on the market.
- Revalidation: The validation is based on a local evaluation of doctors’ practice through appraisal.
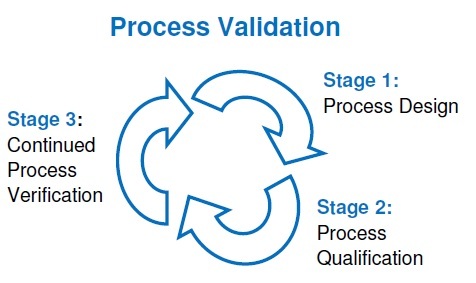
Stages of process validation
1. Stage 1: Process Design
Based on the knowledge gained from product development, the actual manufacturing scale will be determined at this steps. Some factors is necessary to identify includes:
- Quality Target Product Profile (QTPP)
- Critical Quality Attributes
- Critical Process Parameters
- Peform risk assessment
- Create a strategy for process control
2. Stage 2: Process Assessment
This phase evaluates the previously designed process to ensure it can reproduce consistent and reliable levels of quality.
3. Stage 3: On-going process validation
Validation is conducted during the production of commercial batches to ensure that the designed process is capable of satisfying the requirements for quality, stability, and consistency. Furthermore, potential problems that were not detected in previous stages may be discovered and resolved.
It is important that all documents related to unusual events that may affect the quality of the product be recorded and stored.
REFERENCES: