PEARLITOL® SD MANNITOL
In designing tablet products, formulation scientists always look for diluents with good compressibility and chemical inertness toward active pharmaceutical ingredients (APIs). Compared to common fillers such as cellulose derivatives, lactose, or calcium diphosphate, mannitol emerges as an attractive option for both pharmaceutical and nutraceutical applications.
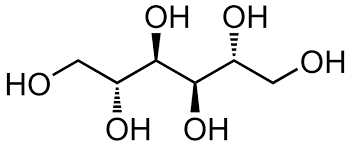
The chemical structure of D-Mannitol
Mannitol (also known as D-Mannitol), a polyol, is commonly used in the pharmaceutical industry. It exists as white or almost white crystalline powder/granule. The PEARLITOL® SD mannitol range is well known across the pharmaceutical industry thanks to its remarkable inertness and stability – making it compatible with most drugs and oral dosage forms. Different from cellulose derivatives, which are usually tasteless and gritty in the mouth, mannitol is slightly sweet, freely water-soluble, and pleasantly cool. Therefore, mannitol is an excellent candidate for chewable tablets, lozenges, and ODTs. Other advantages of mannitol are its low moisture content and low hygroscopicity (absorbing less than 5% of moisture at humidity as high as 90%), which are critical in stabilizing moisture-sensitive APIs. To optimize this excipient for the direct compression process, Roquette has introduced the series PEARLITOL® SD MANNITOL.
Advantages of PEARLITOL® SD MANNITOL
Stability
- Low moisture content even at high relative humidities. Suitable for unstable APIs, especially those sensitive to moisture.
- PEARLITOL® 150 SD with low reducing sugar content – less than 300 ppm – suitable for APIs sensitive to reducing sugars (such as galactose, glucose, glyceraldehyde, fructose, ribose, and xylose, and lactose with the risk of Maillard reaction).
- Promises tablet robustness and quality preserving stability of the formulation over time (even at high temperatures).
Consistent quality & Global supply
- Low batch-to-batch variation ensuring reproducible product and high-performance batch after batch.
- Pearlitol Mannitol is available in Versatile particle size ranges to adapt to any API formulation requirements. Average particle sizes in PEARLITOL® Mannitol 100 SD, 150 SD, and 200 SD is available in the increase in the same order.
- Documentation package enabling Quality by Design.
Superior, reliable functionality
- Optimal for direct compression, which cuts production expenses and increases productivity significantly.
- Absorbing heat during dissolution, it imparts a gently cool sensation when taken orally.
- High compatibility with varying tablet shapes and processes, enabling faster formulation development.
- Low-speed sensitivity and improved compressibility, maximizing productivity and scalability.
- Lower maintenance costs and energy-saving.
Patient Compliance:Sugar-free and non-cariogenic properties despite its slightly sweet taste. It is fit for populations including pediatric and diabetic.
Technical and regulatory supporting documentation is fully available: Conformance to requirements in EP/USP-NF/JP/CHP standards, and availability of many documents, such as DMF, CEP, EXCiPACT, Halal, Kosher, BSE, and Vegan.
Comparison of variants in the PEARLITOL® SD MANNITOL
| 100 SD | 150 SD | 200 SD | |
| Method of manufacture | Spray drying | ||
| Appearance | White to almost white crystalline powder | ||
| Solubility | Freely soluble in water, sparingly soluble in 95% ethanol, practically insoluble in ether. | ||
| Melting temperature | 166 – 170 °C | ||
| Maximum water content (LOD) | 0,5 % | ||
| Average particle diameter | 100 µm | 150 µm | 200 μm |
| Particle size distribution by Laser diffraction |
dv10 = 20 µm dv50 = 100 µm dv90 = 185 µm |
dv10 = 60 µm dv50 = 115 µm dv90 =185 µm |
dv10 = 65 µm dv50 = 150 µm dv90 = 240 µm |
| Bulk density | 0.47 g/cm3 | 0.46 g/cm3 | 0.48 g/cm3 |
| Tapped density | 0.57 g/cm3 | 0.55 g/cm3 | 0.57 g/cm3 |
Application of PEARLITOL® SD MANNITOL
The PEARLITOL® SD MANNITOL series has a wide range of applications in pharmaceutical and nutraceutical manufacture:
- Act as a filler/ diluent for direct compression of chewable tablets, lozenges, effervescent tablets, dispersible tablets, and ODTs due to its mild sweetness, cool feeling, and great organoleptic property
- Act as a filler/ diluent for direct compression tablets due to its exceptional physicochemical stability and no hygroscopicity. Particularly suitable for moisture-sensitive active ingredients such as Vitamins
- Learn more at: Roquette Pharma Virtual Lab
About Roquette and Brenntag
Roquette is a global leader in plant-based ingredients and a top supplier of pharmaceutical excipients. The company now operates in more than 100 countries and with a turnover of about 3.5 billion euros.
In Vietnam, PEARLITOL® SD MANNITOL (manufactured by Roquette) is distributed by Brenntag, which is a leading company in the global distribution market of excipients and APIs.
REFERENCES:
- [1] Roquette expands PEARLITOL® SD mannitol range for APIs
- [2] PEARLITOL Mannitol SD from Roquette Pharma – by American Pharmaceutical Review
- [3] Product profile – PEARLITOL® 100 SD
- [4] Product profile – PEARLITOL® 150 SD
- [5] Product profile – PEARLITOL® 200 SD
- [6] Specifications sheet for PEARLITOL® 100 SD
- [7] Specifications sheet for PEARLITOL® 150 SD
- [8] Specifications sheet for PEARLITOL® 200 SD




